- Pigmentation
- Fine Lines
- Wrinkles
- Sagging Skin
- Rosacea
So far so good!
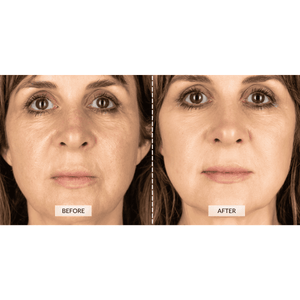
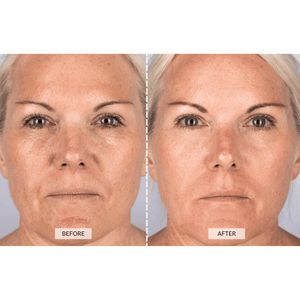
I have done the 8 week starting regime and have done a before and after comparison shot. It is one week after completing the starting set. My eyebrows appear to have lifted and while doing so have imporoved my sagging eyelids!
The yellowish pic was taken on Feb 21st and the second photo on April 21st!
As it is only early days, and it takes up to six months to see the final results I am gobsmacked already!
This machine was a worthy investment and well worth the money. Thank you Currentbody RF!
| Quality |
Rated 5 out of 5
|
|---|---|
| Results |
Rated 5 out of 5
|
| Ease of Use |
Rated 5 out of 5
|